Single Photon Emission Computed Tomography (SPECT) is a nuclear medicine imaging technique, generating 3D images of radiopharmaceutical distribution that elucidate the function of tissues and organs. This technique involves administering a gamma ray-emitting radioisotope, typically through an intravenous injection. The radioisotope can either be a straightforward soluble ion, like gallium (III), or more frequently, it's attached to a specific ligand to create a radioligand with affinity for particular tissue types. Once administered, this compound travels and localizes within specific areas of the body, allowing for targeted imaging via a gamma camera.
Beyond the capabilities of traditional gamma cameras, which are limited to planar 2D imaging, SPECT produces enhanced resolution by accurately locating the tracer in three dimensions. This precision is critical when navigating intricate anatomical landscapes, for example, in discerning reduced function in the heart's inferior wall or pinpointing the exact location of a tumor in the thoracic cavity.
To create a 3D SPECT image, the process includes a complicated mathematical reconstruction of images captured by a gamma camera that rotates around the patient, detecting the emitted gamma rays following tracer decay. Compiled from various angles, these images are reconstructed by computer reconstruction algorithms into a detailed 3D representation. By providing such depth of detail, SPECT substantially improves the diagnostic and monitoring process for a range of medical conditions, from heart disease and cancer to thyroid irregularities, ultimately guiding more tailored and efficacious treatment regimens.
History
The term "Single Photon" in SPECT was originally coined to differentiate it from Positron Emission Tomography (PET), which localizes events using two photons and requires distinct equipment. SPECT, therefore, became the designated term for the 3D imaging of radiopharmaceuticals emitting single photons rather than positrons.
SPECT's history dates back to 1963 when Kuhl and Edwards introduced a pioneering method for nuclear tomographic imaging [1]. Their invention involved a custom-designed scanner that, along with a computational technique known as simple back-projection, could create 3D images. This innovation preceded the work of Hounsfield on CT imaging by approximately a decade.
Kuhl and Edwards continued their advancements by developing the Mark IV scanner, a specialized SPECT device for brain imaging [2]. Throughout the 1970s, brain imaging remained the focus of several dedicated SPECT devices, yet research was simultaneously exploring how to adapt the existing gamma camera—common in all nuclear imaging applications—for SPECT use. This adaptation required the gamma camera to encircle the patient, capturing images from different angles.
Keyes et al. made a significant breakthrough by constructing the first prototype of a rotating gamma camera [3]. This was achieved by repurposing the gantry of a decommissioned cesium-137 radiation therapy unit. Meantime, Jaszczak et al. brought to market a commercial rotating gamma camera model [4].
As dedicated SPECT devices continued to advance, rotating gamma cameras became the primary choice in clinical settings due to their versatility and widespread availability.
SPECT Data Acquisition and Processing
In SPECT, data acquisition is a systematic process where a set of images—termed projections—are captured at multiple, evenly spaced angles around the patient. The patient is positioned so that the axis of rotation for these images aligns with their longitudinal body axis, creating a circular arc as depicted in imaging diagrams (Figure). Typically, these projections span either a complete 360-degree rotation or a half-circle of 180 degrees, with the number of necessary projections for adequate angular sampling ranging from 50 to 150. This range varies depending on the size of the imaged area and the specific spatial resolution capabilities of the SPECT system in use.
The gamma camera's role is crucial; it is fitted with a parallel-hole collimator that ensures photons from the patient are directed perpendicularly towards the camera's detector. This configuration guarantees that the trajectory of the photons, or the "line of origin," can be tracked back from the detector to the source within the patient, forming the basis for image construction.
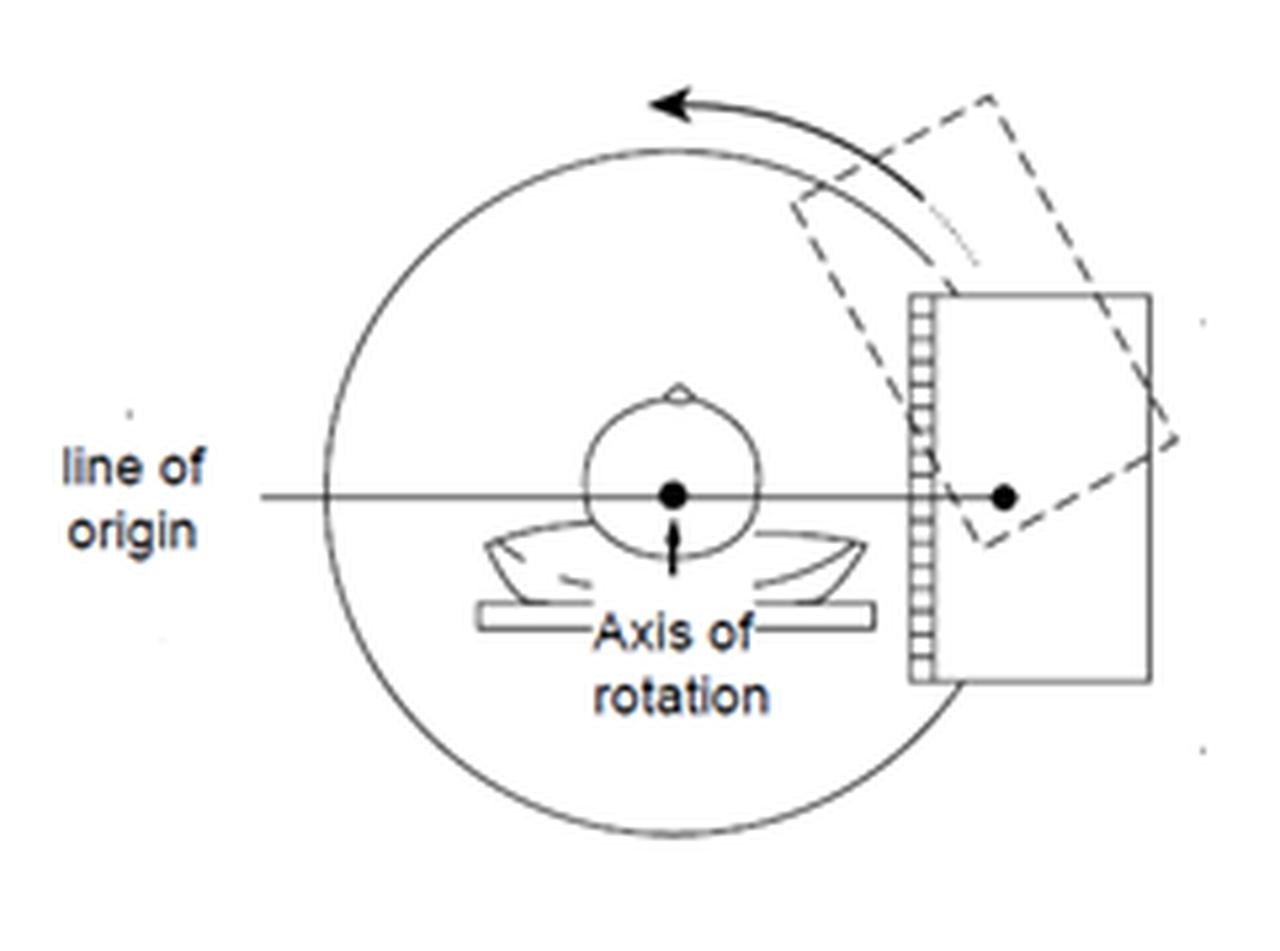
For SPECT data acquisition, the gamma camera rotates acquiring projection images at different angles about the patient. These data are subsequently reconstructed into cross-sectional slices that indicate the 3D in vivo distribution of the radiopharmaceutical within the patient.
Imagine a scenario where we're visualizing a small, contrast-rich feature like a lesion within a cylindrical phantom containing a radioactive substance. A few projections can be taken—three for simplicity in our example. Although the number is limited, if we slice through the phantom's central axis, each projection reveals a consistent intensity due to the lesion's presence. The reconstructions of these projections into a composite image, a process known as "tomographic reconstruction," employ a method called "filtered back-projection (FBP)." This method mathematically processes and integrates the projections, aligning them according to the angle they were taken. The result is a single, synthesized image that represents the object's internal distribution of radioactivity.
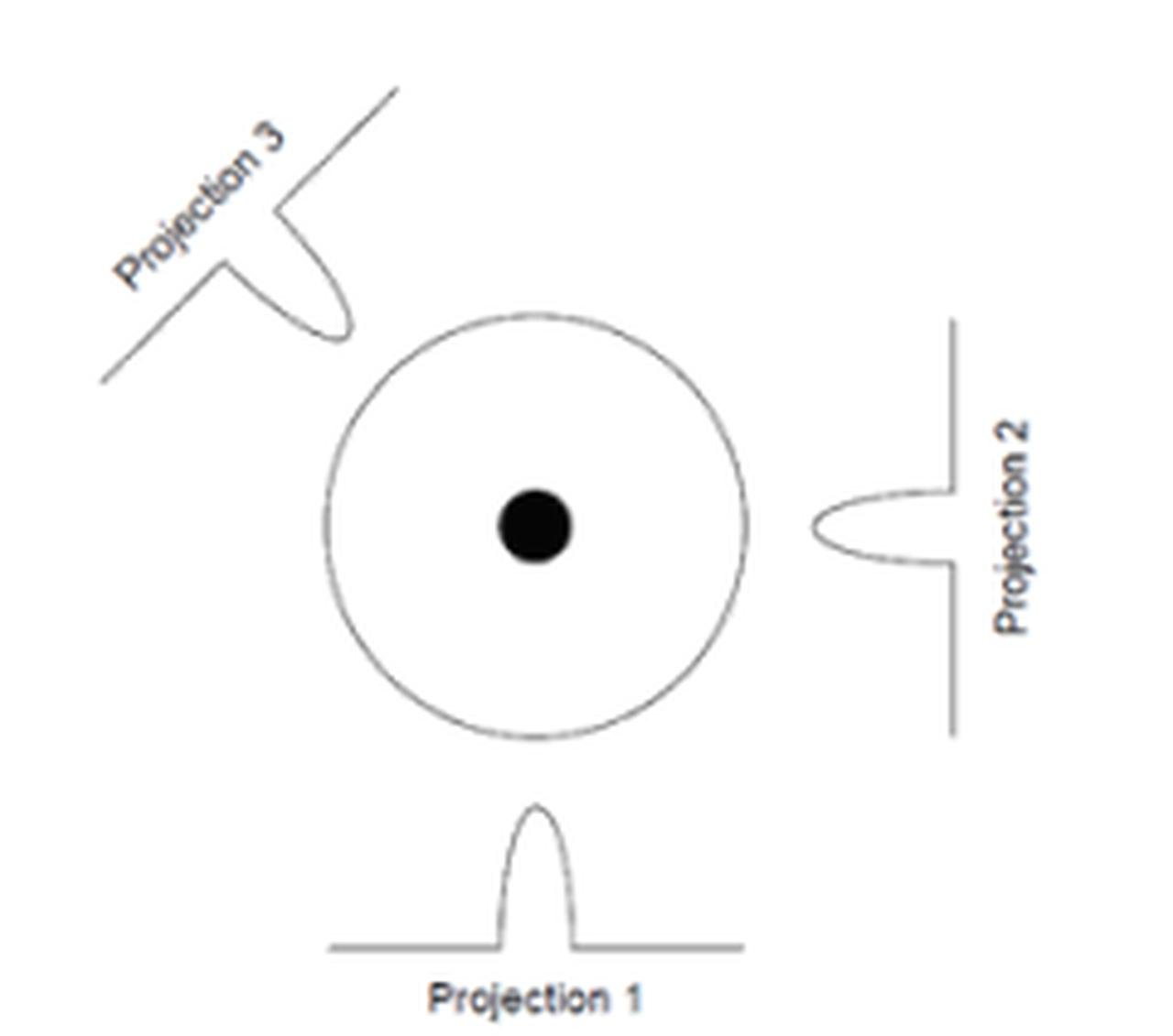
The object in the middle has a central region of high intensity. Three projections about the object are also shown. For a typical SPECT study, 50–150 projections are acquired about the object over 180 or 360 degrees.
FBP typically uses windowing functions—like Hanning, Hamming, Shepp-Logan, and Butterworth filters—to modulate the reconstructed image's sharpness and noise levels. The choice of filter depends on the desired image quality and the characteristics of the initial projection data, impacting the final image significantly. Different clinical scenarios, whether imaging the brain or the heart, may require specific filters to optimize image quality.
While FBP has been traditional in SPECT reconstruction, the advent of iterative reconstruction (IR) methods on newer systems has brought a paradigm shift. These iterative methods start with a guess—an initial approximation of the distribution within the object—and successively refine this estimate. They do so by comparing the calculated projections from the estimate with the actual acquired projections and then adjusting the estimate based on the differences. The iterative process is repeated until the reconstructed image converges to a close representation of the true radioactivity distribution. These methods are advantageous as they can incorporate complex models of the physics of data acquisition and the statistical properties of the detected noise, yielding more accurate reconstructions.
Correcting for attenuation—where deeper tissues emit photons that are more likely to be absorbed before reaching the detector—is critical for accurate quantification in SPECT. Two approaches exist: uniform and non-uniform attenuation correction. The former assumes a constant tissue density and is appropriate for areas like the brain, while the latter is required for heterogeneous areas like the thorax, where tissues of varying densities (e.g., lung, bone, and soft tissue) absorb photons differently.
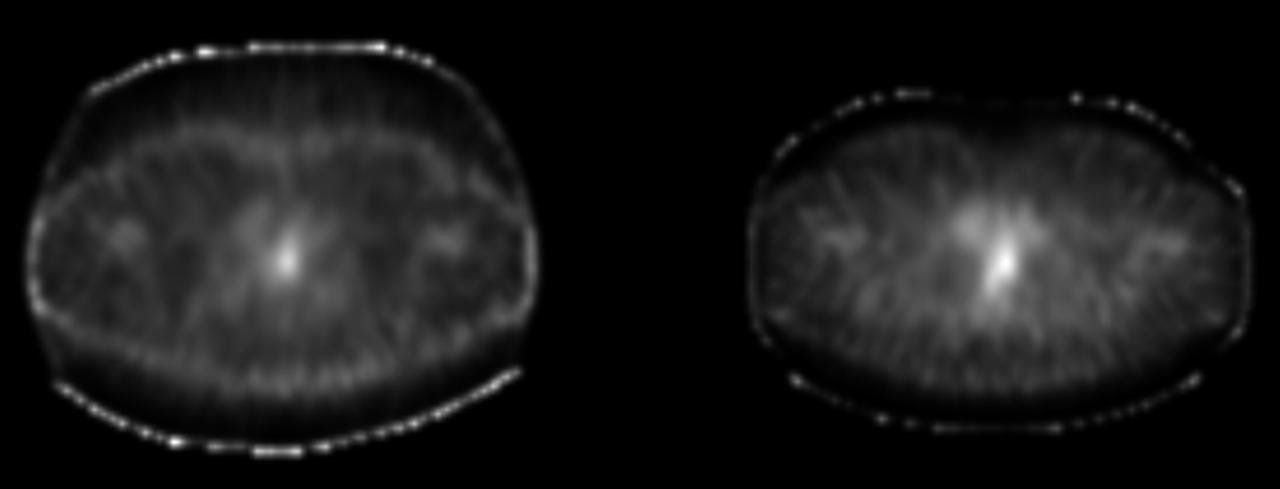
SPECT transverse image without (Left) and with (Right) attenuation correction
Non-uniform attenuation correction has evolved considerably over the past decade. It requires not only the identification of the body's outline but also a detailed understanding of the internal tissue composition, achieved by creating transmission images with an external photon source. These images guide the reconstruction process, compensating for the variable attenuation and improving the fidelity of the SPECT images.
Recent research in the field of SPECT imaging has been geared towards advancing reconstruction and correction methods to improve speed and efficiency. While traditional iterative methods have been enhanced with faster computing and algorithmic innovations, AI-based techniques are emerging as a promising area of exploration. Currently at the forefront of research, these AI methodologies show potential for integration into clinical settings in the future.
SPECT Instrumentation
SPECT requires capturing projection images from multiple angles around the patient. This requires the gamma camera to be affixed to a gantry capable of rotating in a circular or elliptical path around the patient. A notable constraint of this rotating gamma camera system is its low sensitivity. The need for absorptive collimation to ascertain the trajectory of the interacting photons means that only a minimal portion of the photons released by the patient are detected by the camera. Although using tighter collimation can enhance spatial resolution, it does so at the expense of sensitivity, resulting in increased noise in the image.
To enhance sensitivity, a straightforward method is to increase the number of detectors. This approach has led to the development of dual-detector and triple-detector SPECT systems. A modern D-SPECT system feature either 6 or 9 detector columns. The innovative design of Spectrum Dynamics’ D-SPECT camera incorporates patented swivel head detectors. These detectors are pixelated and are aligned with parallel hole tungsten collimators, facilitating faster acquisition times.

A Modern dual-detector with the detectors in a 180-degree opposed arrangement (Left) or at a 90-degree angle (Middle) and A D-SPECT system.
Considering that the heart is situated in the chest's left anterior section and low-energy radiopharmaceuticals like thallium-201 are commonly used, cardiac SPECT data is often collected over a 180-degree arc rather than 360 degrees. The data from the right posterior projection typically contributes only noise and diminished resolution. Consequently, the use of two opposing detectors does not decrease the total imaging duration since a 180-degree gantry rotation is still necessary. To address this, manufacturers have designed dual-detector cameras that can capture data with the detectors in a 180-degree opposed arrangement or at a 90-degree angle as depicted in the figure of SPECT systems, allowing for the collection of the same SPECT data in half the usual time. Adding a third detector further enhances the sensitivity of the SPECT apparatus. Triple-detector systems are highly efficient in SPECT imaging but are less versatile for other nuclear imaging applications, making them less favored than dual-detector systems.
For accurate non-uniform attenuation correction, a transmission scan is essential. Various methods for obtaining this transmission image have been established. One approach uses collimated, radioactive line sources scanned over the patient concurrently with the emission data collection. The radionuclide in these sources (153Gd) emits gamma rays of a slightly different energy than the administered radiopharmaceutical, permitting simultaneous acquisition of both emission and transmission datasets. Alternatively, multiple smaller line sources with 153Gd are utilized for the same purpose.
Today, hybrid SPECT–CT systems have been widely employed. These combine a helical CT scan with a SPECT scan in a single device. The CT scan aids in characterizing the internal materials for a precise non-uniform attenuation correction, despite requiring a conversion between CT values and SPECT attenuation coefficients due to differing photon energies. In some instances, these systems can yield diagnostic-quality CT images that can be analyzed alongside or separate from the SPECT results. However, in one type of SPECT–CT system, the CT is of non-diagnostic quality and is solely used for attenuation correction and basic anatomical reference.
Advances in molecular medicine have sparked interest in imaging small animals like rodents, leading to the development of SPECT imaging techniques with very high spatial resolution. One direct approach employs tiny pinhole collimators, around 1mm in size, much smaller than the 4–6mm typically used in clinical settings. This can produce SPECT images with spatial resolution nearly ten times finer than standard clinical SPECT. However, these high-resolution methods are impractical for clinical use since the tiny pinholes significantly reduce efficiency, requiring impractically long imaging times to achieve human images with adequate quality.
Clinical Applications
The capability of SPECT to pinpoint locations within a 3D space makes it particularly useful for assessing localized functions within internal organs. This feature is especially beneficial for detailed functional imaging of the heart or brain, where understanding the spatial dynamics of organ function is crucial.
Cardiology
SPECT's most frequent use in a clinical setting is for the imaging of myocardial perfusion. This method is crucial for identifying atherosclerotic heart disease, which is characterized by the constriction of coronary arteries, restricting blood flow to the heart and potentially leading to chest pain or myocardial infarction. SPECT serves to either confirm or rule out substantial coronary artery disease. If SPECT results are abnormal, further invasive diagnostic procedures, such as coronary angiography, might be pursued, possibly leading to interventions like angioplasty or bypass surgery.
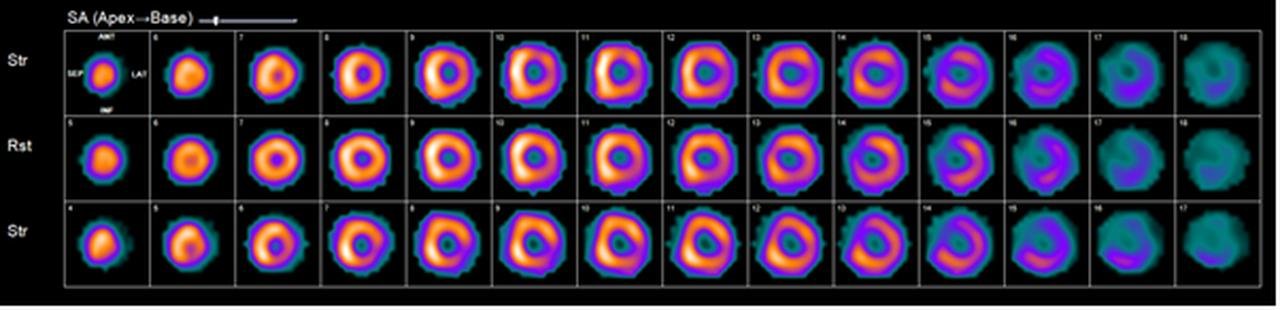
SPECT Myocardial Perfusion Imaging (MPI)
For imaging purposes, radiopharmaceuticals like thallium-201 or more modern technetium-labeled compounds are introduced into the bloodstream, where they are taken up by the myocardium. The SPECT images produced provide a three-dimensional representation of blood distribution to the heart muscle as delivered by the coronary arteries.
The procedure entails two phases: one during rest and the other during induced stress, typically through treadmill exercise, although medication-induced stress is an alternative for those unable to undergo physical exertion. In a healthy heart, SPECT will show robust blood flow in both stages. While in hearts that have experienced a myocardial infarction, SPECT will reveal no blood flow in the affected region during either phase. Patients with significant coronary artery stenosis will display normal resting images but abnormal stress-induced images due to the inability of the obstructed artery to meet the heightened demand for oxygen and blood during stress.
Oncology
SPECT, paired with various radiopharmaceuticals, provides crucial information on the extent and distribution of cancer within the body. This data is integral for initial diagnosis, staging, surgical planning, and assessment of treatment response.
For decades, Gallium-67 citrate (67Ga) has been a staple in tumor imaging, with nonspecific binding properties across a range of tumors. Gallium-67 SPECT is primarily employed for imaging malignant lymphoma and is more accurate than CT or MRI in evaluating the efficacy of therapy.
For imaging neuroendocrine tumors (like carcinoid, gastrinoma, and others), 111In OctreoScan is utilized due to its affinity for somatostatin receptors, often revealing small tumors that may go undetected by CT or MRI.
111In ProstaScint, a radiolabeled monoclonal antibody targeting prostate cancer cell antigens, enables the identification of recurrent prostate cancer sites, which may be indicated by rising PSA levels. SPECT is essential here due to the high background activity that otherwise hampers imaging.
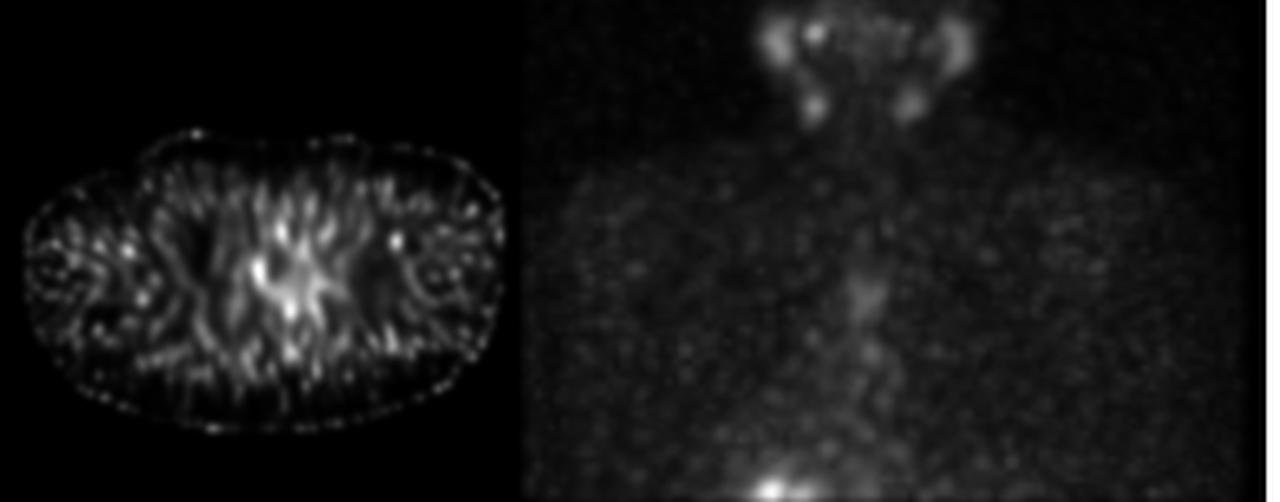
NM tumor scan with SPECT (Thyroid with NaI)
Neurology
In brain imaging, SPECT is indispensable for visualizing the intricate overlay of the brain's convoluted regions.
SPECT is utilized in the localization of seizure foci for patients whose seizures are unresponsive to medication. While EEGs can be moderately effective, they often require confirmation through additional tests. Traditional methods involve invasive electrode placement on the brain's surface, which carries risks. Radiopharmaceuticals like 99mTc HMPAO and 99mTc ECD used in SPECT show blood flow distribution in the brain, highlighting areas of increased metabolism and blood flow during seizures, and decreased activity between seizures, allowing for precise localization of seizure origins.
SPECT also plays a role in diagnosing causes of dementia, with distinctive perfusion patterns evident in conditions like Alzheimer's disease, frontal lobe dementia, and dementia resulting from multiple infarctions (strokes).
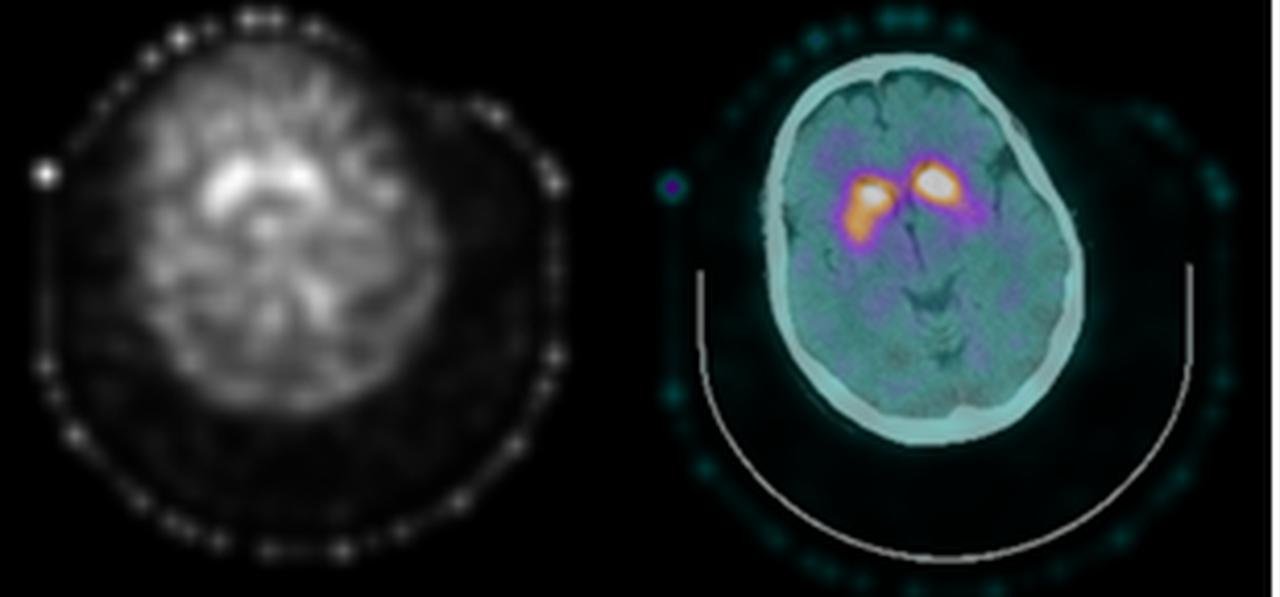
NM Brain Scan with SPECT/CT (5.7mCi of I-123 ioflupane (DaTscan). Normal, not consistent with Parkinson’s disease or other presynaptic parkinsonian conditions.
Orthopedics
SPECT is valuable for detecting subtle instances of tumors, fractures, or infections that may be difficult to discern with traditional two-dimensional bone scans. It aids in precisely locating bone radiopharmaceutical uptake, distinguishing benign from malignant processes, confirming the sources of pain due to small fractures, and identifying infection sites.
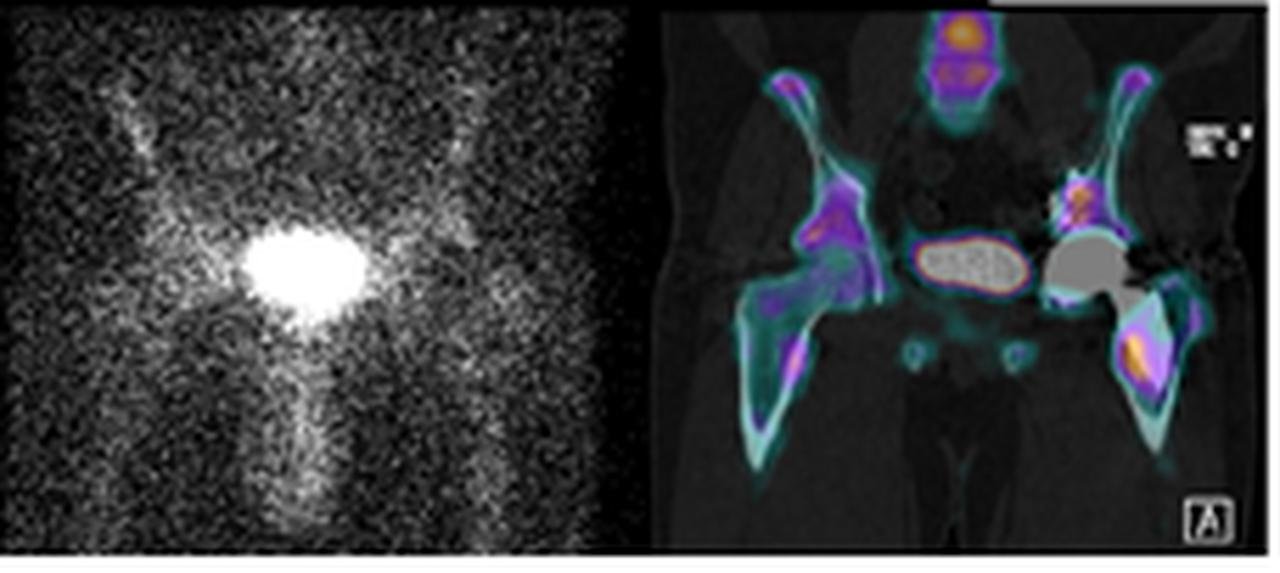
NM Bone with SPECT/CT. Left: SPECT image; Right: SPECT-CT fused image
Reference
- Kuhl, D.E. and R.Q. Edwards, Image Separation Radioisotope Scanning. Radiology, 1963. 80(4): p. 653-662.
- Kuhl, D.E. and R.Q. Edwards, The Mark 3 Scanner: a compact device for multiple-view and section scanning of the brain. Radiology, 1970. 96(3): p. 563-70.
- Keyes, J.W., Jr., et al., The Humongotron--a scintillation-camera transaxial tomograph. J Nucl Med, 1977. 18(4): p. 381-7.
- Jaszczak, R.J., et al., Radionuclide emission computed tomography of the head with 99mTc and a scintillation camera. J Nucl Med, 1977. 18(4): p. 373-80.