A gamma camera, also known as a scintillation camera or Anger camera, is a specialized medical imaging device used to detect and visualize gamma radiation emitted by radioactive substances administered to the body, a technique known as scintigraphy. This imaging technique is commonly used in drug development and nuclear medicine imaging to diagnose and monitor a variety of medical conditions.
History
In the early stages of nuclear medicine, during the 1950s, there was a growing interest in utilizing radioactive isotopes for medical imaging and diagnosis. Technological progress enabled the acquisition of images depicting the distribution of radionuclides within the human body, a marked departure from mere point measurements. In 1950, Benedict Cassen introduced rectilinear scanner, which produced images slowly and no capability to image rapidly changing distributions[1]. The rectilinear scanner was eventually replaced by the Anger camera, invented by Hal O. Anger. In 1957, Anger completed his first gamma imaging camera that he called a scintillation camera and is often referred to as the Anger camera [2, 3]. Anger's invention featured sodium iodide scintillation crystals as detectors, emitting bursts of light upon interaction with gamma radiation. This innovation led to a significant enhancement in the camera's sensitivity and efficiency, facilitating precise localization and detection of radiation within the human body. By 1963, he had a system with a 28 cm field of view and 19 photomultiplier tubes. This device became commercialized as the nuclear Chicago scintillation camera.

H.O.Anger and his first gamma camera
Until the early 1960s, nuclear medicine predominantly relied on 131I for the study and diagnosis of thyroid disorders and specific organ-related investigations. About the same time that the Anger camera was introduced, the landscape of nuclear medicine underwent a transformative shift in 1964 when Paul Harper and colleagues [4] introduced 99mTc for imaging. The gamma rays emitted by 99mTc offered superior imaging properties, coupled with unparalleled versatility for labeling a wide array of compounds, allowing the study of nearly every organ in the body. Equally significant, its production in a relatively long-lived generator form facilitated readily available supplies of the radionuclide. Currently, 99mTc stands as the most widely used radionuclide in nuclear medicine. The Anger camera's optimization for 99mTc further enhanced its effectiveness. Hal O. Anger's contributions and developments in radionuclide imaging have significantly advanced the field of nuclear medicine.
As the field of nuclear medicine continued to advance, the 1970s witnessed a range of technological enhancements in gamma camera design. These developments included the introduction of multi-crystal detectors, more sophisticated collimators, and improved image processing techniques. Collectively, these innovations not only boosted the quality of nuclear medicine imaging but also broadened the range of applications for the gamma camera.
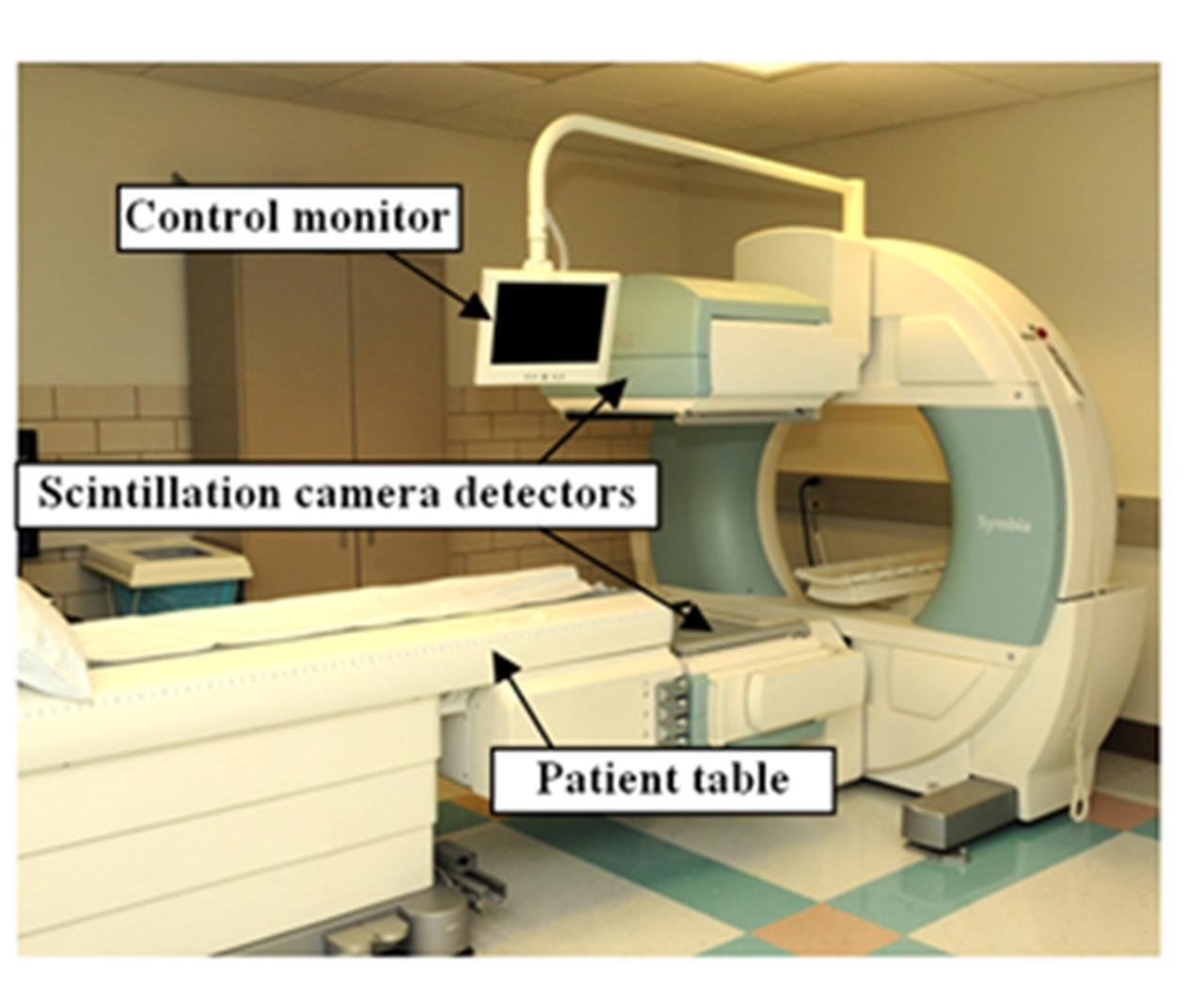
Dual Head Modern Gamma Camera
In the subsequent decades, research and development in the field of nuclear medicine continued to refine and expand the capabilities of the gamma camera. These improvements encompassed detector technology, image processing software, and patient comfort. Today, gamma cameras are indispensable tools for the detection and evaluation of a wide array of medical conditions, including tumors, cardiac function assessment, neurological studies, and assessments of organ function. The gamma camera's remarkable journey, from the early exploration of nuclear medicine to becoming a sophisticated and versatile diagnostic instrument, underlines its profound impact on healthcare and diagnostics.
System Description
The Anger camera is a sophisticated imaging device composed of several key components. Its functionality begins with a large, slender sodium iodide crystal designed to absorb gamma-ray energy and transform it into visible light. This light is then sampled and converted into electronic pulses by an array of photomultiplier tubes (PMTs).
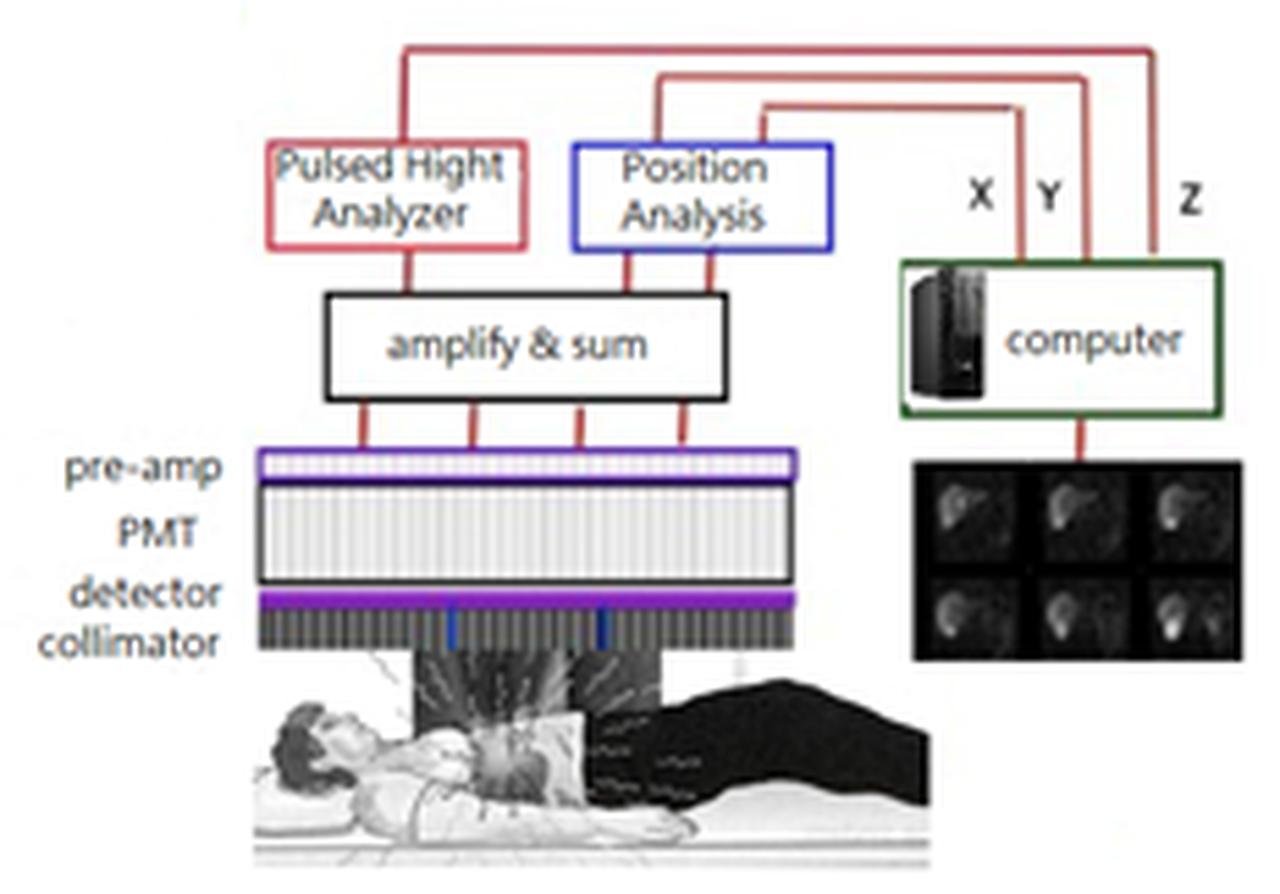
Imaging Chain of Gamma Camera
To create an image of the distribution of radionuclides, the radiation must be projected onto the detector. In traditional cameras, optical lenses are employed for image projection. However, gamma rays are too energetic to be focused using optics or other materials. The initial solution for gamma-ray imaging projection was the pinhole collimator. The pinhole collimator used in an Anger camera closely resembles a conventional pinhole camera. It inverts the object and magnifies or reduces the image based on the ratio between the pinhole-to-detector distance and the object-to-pinhole distance. Pinhole collimators are usually constructed using tungsten and necessitate lead shielding around the "cone." Nevertheless, pinhole imaging of large three-dimensional distributions can result in distortion, and sensitivity to off-axis activity diminishes rapidly. For most imaging scenarios, a more effective solution is a multiholed parallel collimator (see Fig). The parallel collimator consists of numerous holes with lead septa, typically featuring hexagonal holes around 1.5mm in diameter and 20–30mm in length, with septal walls around 0.2 mm thick. The parallel hole collimator generates projections without magnification, allowing gamma rays that pass through the holes to reach the detector, while those with trajectories intersecting the septa are absorbed. Less than 1 out of 5000 gamma rays impacting the front surface of the collimator make it through to the detector, forming the image.
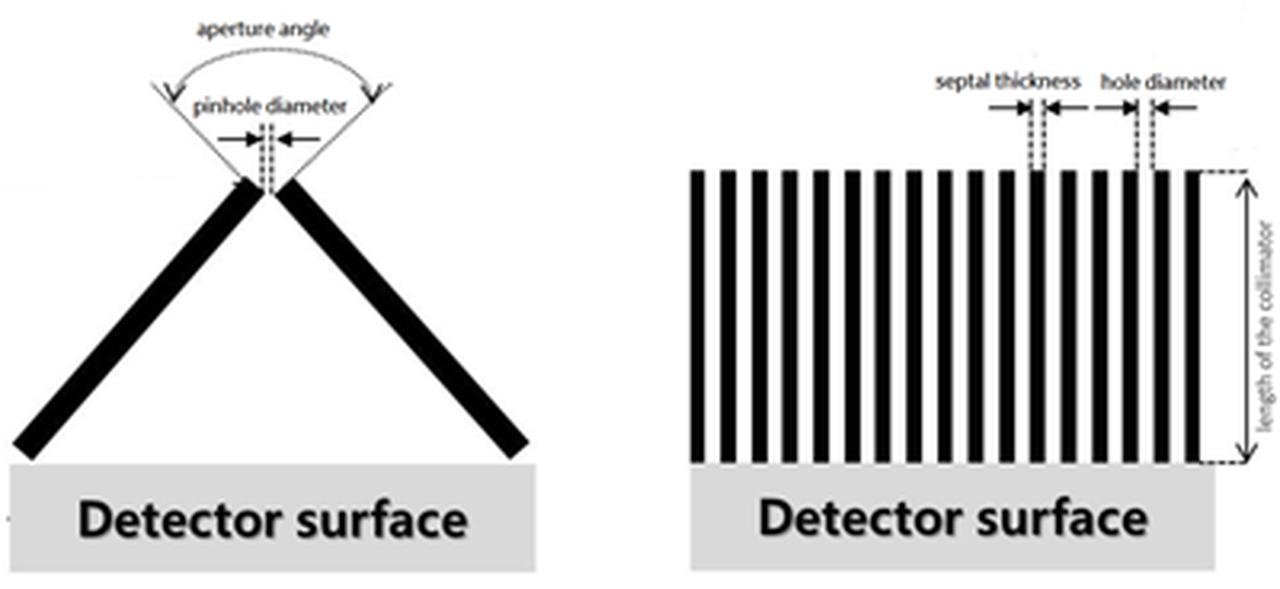
Pinhole Collimator (Left) and Parallel Collimator (Right)
In Anger cameras, the crystals are typically 40-55 cm in size and 9.5 mm thick. Because NaI(Tl) is hygroscopic and absorbs moisture from the air, it must be hermetically sealed. Any breach of this seal typically leads to the crystal turning yellow and becoming irreversibly damaged.
The visible light produced from the absorption of a gamma ray in the NaI (Tl) crystal conveys both location and energy information. The intensity of the scintillation is directly proportional to the energy absorbed during the event. To utilize this information, the scintillation must be converted into an electronic signal, which is achieved through the use of PMTs. In a PMT, scintillation light liberates electrons at the photocathode, and these electrons are then amplified through a series of dynodes. The overall gain provided by a photomultiplier tube is on the order of 106.
Determining the energy of the detected event is crucial. Gamma rays that are entirely absorbed create a scintillation signal directly proportional to the gamma-ray energy. Consequently, the signal resulting from the unweighted sum of the PMTs represents the gamma-ray energy, and this signal is sent to a pulse height analyzer. To reject scattered radiation from within the patient, an energy window is set to select events resulting from the total absorption of the primary gamma ray. Gamma rays that have been scattered in the patient typically lose energy and are predominantly excluded. The position of the gamma-ray event on the detector is determined by summing weighted signals from the PMTs [6-8].
In the past, Anger camera images were produced using photographic techniques, where the X and Y signals intensified a dot on a cathode ray tube (CRT) observed by a camera. However, modern Anger cameras have replaced the CRT with computer memory, and the location information is now in digital form. The X and Y coordinate values, still facilitated by the PHA, point to a specific memory element in a computer matrix. The content of that memory element is incremented by 1, and this process continues, accumulating information in the computer matrix until the count or time-stopping criteria are met.
Spatial Resolution
The use of a collimator to direct gamma-ray photons introduces complexities and attributes that impact image quality and precision. Collimators encompass different types—namely, low energy, medium energy, and high energy variants. These variants, tailored to diverse gamma-ray energy ranges, exhibit varying hole dimensions, thickness, and septations. Notably, as collimators transition from low to high energy, a trade-off arises: enhanced efficiency is counterbalanced by reduced spatial resolution, particularly when the radiation source is situated at a greater distance from the collimator.
Each aperture within the collimator governs the form of detected photons, effectively constraining them within a conical shape. This geometry introduces a degree of image blurring, partly attributed to the fact that lead, the principal collimator material, does not entirely block scattered incident gamma photons. Consequently, this feature may lead to crosstalk or overlap between neighboring apertures, further affecting image clarity.
Moreover, the application of a collimator significantly curtails the camera system's sensitivity. Unlike optical lenses in visible light cameras that transmit most incoming photons, a collimator attenuates a substantial portion of incident gamma photons. Consequently, this limited sensitivity necessitates a high radiation dose to ensure the detection of an adequate number of scintillation points, an imperative for meaningful image creation.
While endeavors have been made to enhance this approach, with explorations into alternatives like pinhole collimators and rotating slat collimators with CZT detectors, their widespread adoption in routine clinical practice remains relatively limited.
The current camera systems can distinguish between two separate point sources of gamma photons at distances ranging from 6 to 12 mm, depending on factors like the collimator type, radioisotope used, and distance from the collimator. However, spatial resolution declines rapidly with increased distance from the camera face. This limitation affects the precision of the computer-generated image, particularly in heart muscle imaging, where the muscle is often more than 5 cm from the collimator and in constant motion. Advanced systems mitigate this by limiting scintillation counting to specific phases of the heart's contraction cycle, known as gating, although this reduces system sensitivity.
Applications
1. Bone Scintigraphy: Gamma cameras can detect abnormalities in bone, such as fractures, infections, or bone cancer. They are valuable tools for assessing bone health and identifying bone-related conditions.
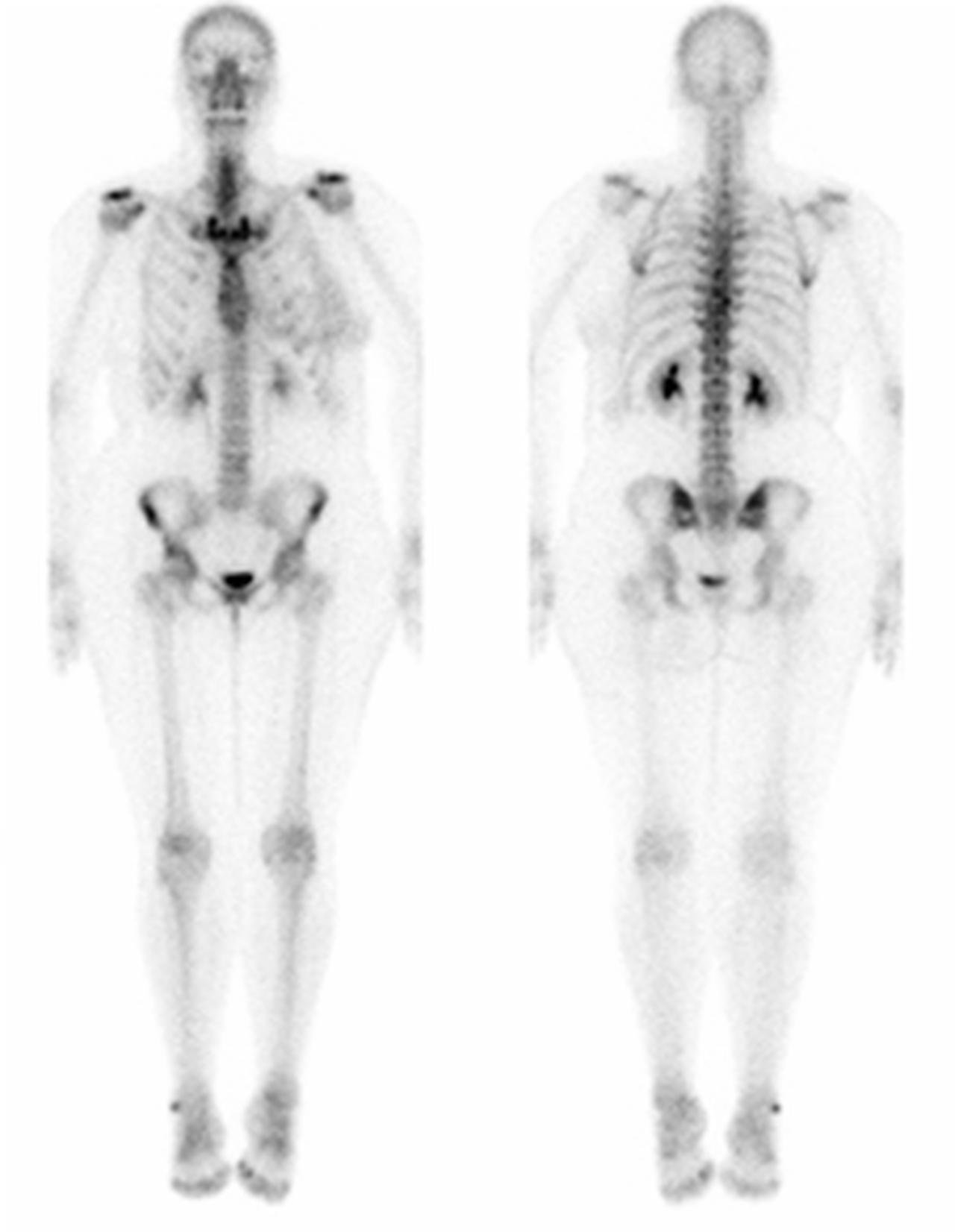
Whole-body Skeletal Nuclear Imaging to Evaluate Metastatic Spread of Cancer
2. Thyroid Imaging: Gamma cameras are used to assess the function and structure of the thyroid gland. They can detect thyroid nodules, inflammation, and thyroid cancer.
3. Renal Scintigraphy: This procedure is used to assess kidney function, detect obstructions in the urinary tract, and evaluate the blood flow to the kidneys.
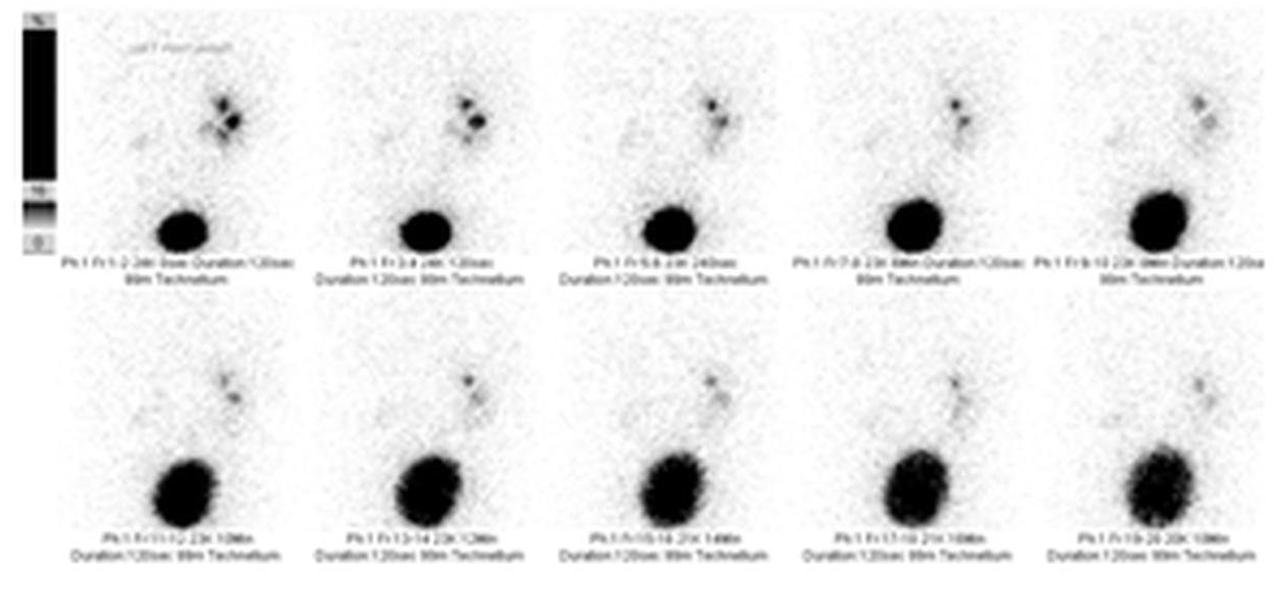
Renal Perfusion and Function Scan
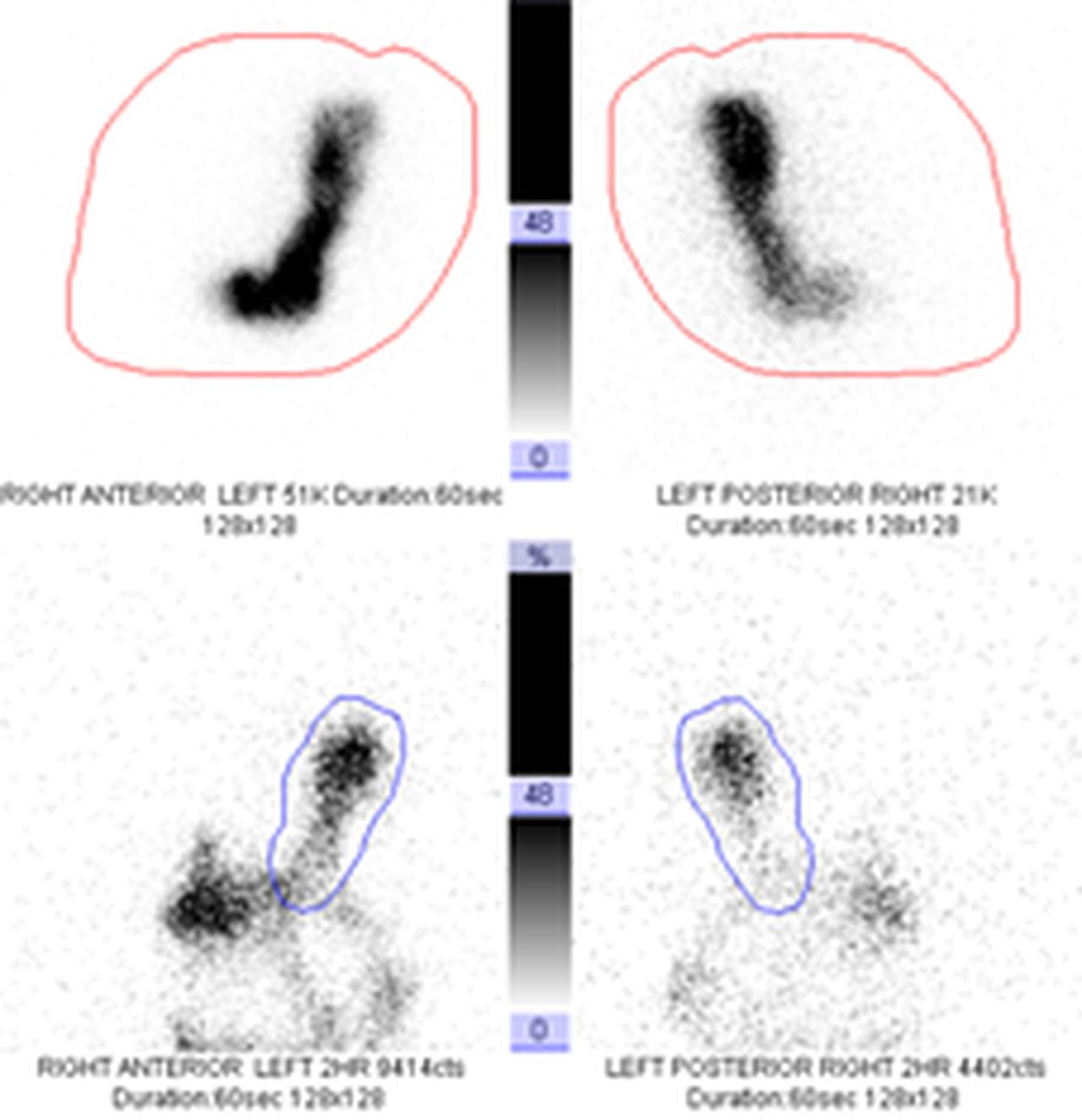
4. Sentinel Lymph Node Mapping: In cancer surgery, gamma cameras help locate and map sentinel lymph nodes, which are the first lymph nodes to which cancer cells are likely to spread.
5. Gastrointestinal Bleed Scans: Gamma cameras can be used to detect and locate gastrointestinal bleeding sites, helping physicians identify the source of bleeding.
Gastric Emptying Scan
6. Pulmonary Scintigraphy: This technique is used to evaluate lung function and perfusion. It can help diagnose pulmonary embolism and assess lung diseases.
7. Brain Imaging: In some cases, gamma cameras are used in brain studies to detect abnormalities, such as tumors, lesions, or areas of abnormal blood flow.
Gamma cameras are powerful diagnostic tools in nuclear medicine, providing valuable information about the function and structure of various organs and tissues. They are especially useful in cases where other imaging modalities, such as X-ray or CT scans, may not provide the required information.
References
1. B Cassen, L.C., C. REED, R. LIBBY, L Curtis-Snell, C Reed, R. T. Libby Instrumentation for 131I use in medical studies. Nucleonics, 1951. 9: p. 46-50.
2. Anger, H.O., Scintillation Camera. Review of Scientific Instruments, 1958. 29(1): p. 27-33.
3. Anger, H.O., Use of a Gamma-Ray Pinhole Camera for in vivo Studies. Nature, 1952. 170(4318): p. 200-201.
4. Harper, P.V., et al. OPTIMIZATION OF A SCANNING METHOD USING Tc$sup 99$m. 1964.
5. Murphy, P.H. and J.A. Burdine, Large-field-of-view (LFOV) scintillation cameras. Seminars in Nuclear Medicine, 1977. 7(4): p. 305-313.
6. Anger, H.O., SCINTILLATION CAMERA WITH MULTICHANNEL COLLIMATORS. J Nucl Med, 1964. 5: p. 515-31.
7. Scrimger, J.W. and R.G. Baker, Investigation of light distribution from scintillations in a gamma camera crystal. Phys Med Biol, 1967. 12(1): p. 101-3.
8. Simon R. Cherry, J.A.S., Michael E. Phelps, Physics in Nuclear Medicine, 4th edition. 2012: Elsevier.